The director of the U.S. Centers for Disease Control and Prevention (CDC) on Tuesday backed broad use of Pfizer's (PFE.N) and BioNTech's COVID-19 vaccine in children ages 5 to 11, clearing the way for shots to go into young arms as soon as Wednesday, Reuters reports.
US Gives Final Clearance to COVID-19 Shots for Kids 5 to 11
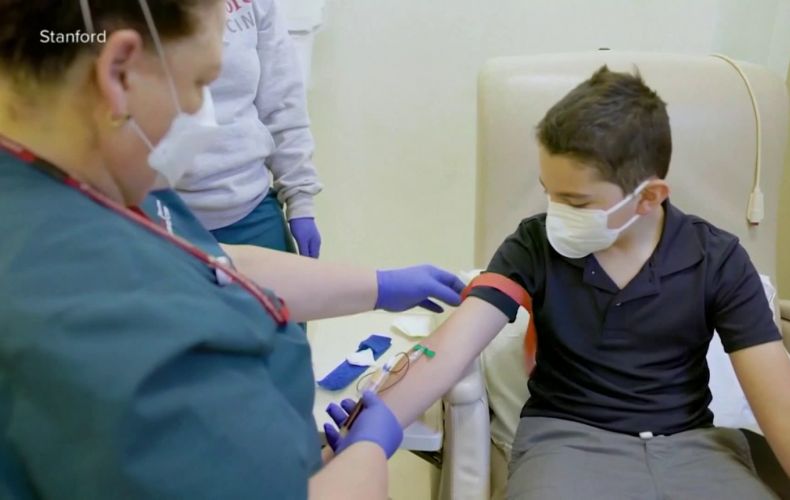
STEPANAKERT, NOVEMBER 3, ARTSAKHPRESS: The U.S. Food and Drug Administration granted emergency use authorization of the vaccine in 5- to 11-year-olds on Friday.
The FDA authorized a 10-microgram dose of Pfizer's vaccine in young children. The original shot given to those age 12 and older is 30 micrograms.